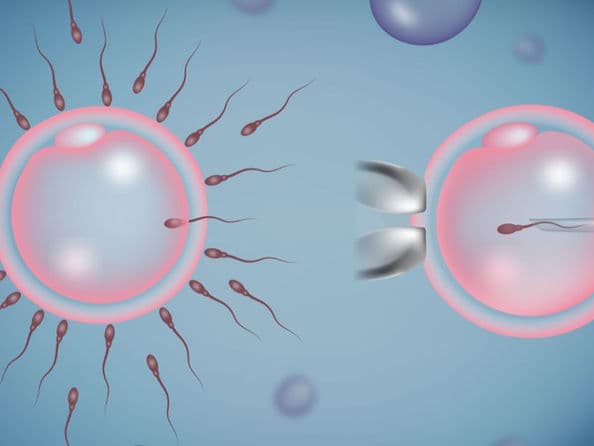
IVF with Intracytoplasmic Sperm Injection (ICSI)
IVF with ICSI Treatment
In Vitro Fertilization (IVF) with Intracytoplasmic Sperm Injection (ICSI) is an advanced laboratory procedure designed to maximize the chances of fertilization by directly assisting the sperm's entry into the egg. In 2026, ICSI has become the gold standard for treating severe male factor infertility and overcoming barriers like hardened egg shells. By utilizing AI-driven sperm selection and microfluidic technology, this procedure ensures that a single, high-quality sperm is precisely injected into a mature egg to facilitate successful embryo development.
When You Should Consider ICSI
Diagnosis of low sperm count (oligospermia) or poor sperm motility (asthenozoospermia).
Presence of high numbers of abnormally shaped sperm (teratozoospermia).
History of failed or poor fertilization in previous traditional IVF cycles.
Use of eggs that have been previously frozen and thawed for a "delayed" cycle.
Requirement for sperm collected through surgical retrieval (TESE/TESA).
Couples undergoing Preimplantation Genetic Testing (PGT) who require precise fertilization data.
Conditions That Require Specialized Care
Severe Male Factor Infertility involving blockages or non-obstructive azoospermia.
Advanced Maternal Age where the egg's outer shell (zona pellucida) may be thicker or harder.
Unexplained Infertility where traditional fertilization methods have been unsuccessful.
Genetic conditions that require the selection of sperm with the highest DNA integrity.
Cryopreserved egg cycles where the freezing process has altered the egg's membrane.
How IVF with ICSI Is Performed
Ovarian stimulation is used to produce multiple mature eggs, which are then surgically retrieved.
Microfluidic selection devices (Zymot) are utilized to isolate sperm with the highest DNA integrity.
AI-driven imaging analyzes sperm morphology and movement to select the optimal candidate.
The egg is stabilized under a high-powered microscope using a specialized holding pipette.
A single sperm is carefully injected into the center of the egg using a microscopic glass needle.
Fertilized eggs are placed in time-lapse incubators (EmbryoScope) for undisturbed monitoring.
Types of ICSI Innovations in 2026
AI-Driven Sperm Selection Advanced software that identifies the sperm most likely to result in a healthy, viable embryo.
Microfluidic Sperm Sorting A gentle selection method that mimics the natural cervical tract to filter out sperm with DNA damage.
Time-Lapse Embryo Monitoring Continuous 24/7 video observation of the embryo, allowing for grading without removing it from the incubator.
AI Embryo Grading Algorithms that predict "live birth potential" by analyzing thousands of developmental data points.
Non-Invasive Genetic Testing (niPGT) Analyzing the liquid environment around the embryo for chromosomal health, avoiding the need for a biopsy.
Surgical Sperm Retrieval (TESA/TESE) Minor procedures to collect sperm directly from the reproductive tract when it is absent in the ejaculate.
Pre-Procedure Preparation
Complete a comprehensive semen analysis, including DNA fragmentation testing.
Undergo a "mock transfer" to map the uterine path for the eventual embryo implantation.
Adhere to a specialized vitamin and antioxidant protocol to optimize egg and sperm quality.
Manage lifestyle factors such as smoking and alcohol use to improve cellular health.
Participate in a consultation with an embryologist to discuss the specific ICSI plan.
Pre-Procedure Tests
Anti-Müllerian Hormone (AMH) and antral follicle count to assess ovarian reserve.
Sperm DNA Fragmentation Index (DFI) to determine the need for microfluidic selection.
Infectious disease screening for both partners as required by 2026 regulatory standards.
Uterine cavity evaluation (Hysteroscopy or saline ultrasound) to ensure a receptive environment.
Genetic carrier screening to identify any potential risks of inherited conditions.
Why IVF with ICSI Is Highly Effective
Achieves successful fertilization in 70% to 80% of mature eggs injected.
Overcomes severe male infertility barriers that previously made biological parenthood impossible.
Minimizes the risk of "total fertilization failure," providing more embryos for selection.
Utilizes 2026 AI technology to select sperm and embryos with the highest genetic potential.
Offers a standardized solution for patients using frozen-thawed eggs or donor tissue.
Recovery and Monitoring
Minor recovery time following egg retrieval, usually requiring one day of rest.
Monitoring of embryo development via a digital portal provided by the embryology lab.
Administration of progesterone and other hormones to prepare the uterine lining for transfer.
Follow-up consultations to discuss the results of genetic testing or embryo grading.
Emotional support and counseling integrated throughout the waiting period before the pregnancy test.
Life After IVF with ICSI
Transition to prenatal care once a successful pregnancy is confirmed via blood test and ultrasound.
Options for cryopreservation of remaining high-quality embryos for future family building.
Long-term follow-up to monitor the health and development of children born via assisted technology.
Continued management of underlying fertility conditions for future reproductive health.
Engagement with support communities for families who have utilized assisted reproduction